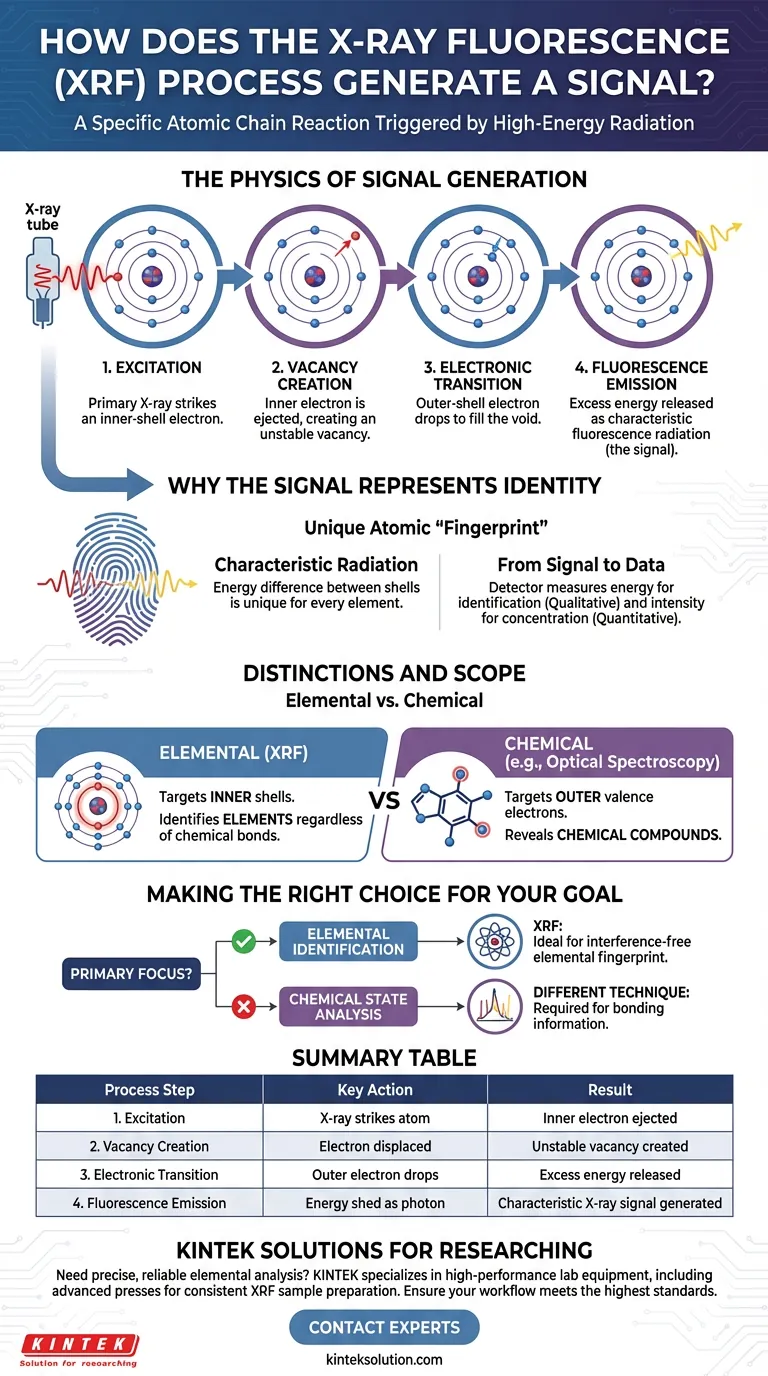
The X-ray Fluorescence (XRF) signal is generated through a specific atomic chain reaction triggered by high-energy radiation. When a primary X-ray strikes a sample, it ejects an electron from an atom's inner shell, creating an unstable vacancy. To restore stability, an electron from a higher-energy outer shell drops down to fill this void, releasing the excess energy as a specific type of light known as fluorescence radiation.
The core principle of XRF is that the signal produced is a direct result of the atom attempting to stabilize itself. Because the energy spacing between electron shells is unique to every element, the emitted fluorescent light acts as a unique "fingerprint" that definitively identifies the element.

The Physics of Signal Generation
To understand XRF, you must look beyond the surface of the material and focus on the interaction between the radiation and the inner electron shells of the atom.
Step 1: The Excitation
The process initiates when an excitation source, typically a primary X-ray from an X-ray tube, bombards the sample material.
This primary X-ray strikes an atom with enough energy to dislodge an electron from its position.
Crucially, XRF targets the inner atomic shell, differentiating it from optical techniques that interact with the outer valence electrons.
Step 2: Creating the Vacancy
When the inner electron is ejected, it leaves behind a vacancy.
This state is energetically unstable for the atom.
The atom immediately seeks to return to a stable state by filling this empty space in its inner structure.
Step 3: The Electronic Transition
To fill the vacancy, an electron from a further outer shell transitions down to the lower energy level.
This outer electron possesses more energy than the inner shell requires.
As the electron drops into the vacancy, it must shed this excess energy to fit the new, lower energy state.
Step 4: Fluorescence Emission
The excess energy is released in the form of a secondary X-ray photon.
This released photon is the fluorescence radiation—the actual signal detected by the XRF instrument.
The intensity of this signal correlates to the concentration of the element within the sample.
Why the Signal Represents Identity
The utility of XRF lies in the specific characteristics of the emitted signal. It is not random noise; it is highly specific data.
Characteristic Radiation
The energy difference between electron shells is fixed and unique for every element in the periodic table.
Therefore, the radiation released during the transition has a characteristic energy specific to that element.
From Signal to Data
The XRF detector captures these photons and measures their energy levels.
By analyzing the specific energy of the fluorescence, the system can identify exactly which elements are present (qualitative analysis).
Simultaneously, by measuring the intensity or amount of this signal, the system calculates the concentration of those elements (quantitative analysis).
Distinctions and Scope
While XRF is a powerful tool for elemental analysis, it is important to understand the specific nature of the signal it generates compared to other methods.
Elemental vs. Chemical
XRF generates signals based on inner shell interactions, which are shielded from chemical bonding environments.
This means XRF is excellent for determining what elements are present (e.g., Iron, Calcium), regardless of how they are chemically bound.
Contrast with Optical Spectroscopy
Optical spectroscopy relies on valence electrons, which are the outer electrons involved in bonding.
Because XRF ignores these valence electrons, it does not typically provide information about chemical compounds or molecular structures, only the elemental composition.
Making the Right Choice for Your Goal
Understanding the source of the XRF signal helps in determining if this is the correct analytical technique for your specific requirements.
- If your primary focus is Elemental Identification: XRF is the ideal choice because the signal relies on inner-shell electron transitions that provide a unique, interference-free fingerprint for each element.
- If your primary focus is Chemical State Analysis: You may require a different technique (like Optical Spectroscopy), as XRF signals do not reflect the valence electron states that determine chemical bonding.
The XRF signal is ultimately a measure of atomic stability, providing a precise and characteristic readout of the fundamental elements composing your material.
Summary Table:
| Process Step | Key Action | Result |
|---|---|---|
| 1. Excitation | Primary X-ray strikes atom | Inner-shell electron is ejected |
| 2. Vacancy Creation | Electron is displaced | Unstable vacancy is created |
| 3. Electronic Transition | Outer-shell electron drops down | Excess energy is released |
| 4. Fluorescence Emission | Energy is shed as a photon | Characteristic X-ray signal is generated |
Need precise, reliable elemental analysis for your laboratory? The XRF process demonstrates the importance of accurate signal generation for identifying material composition. At KINTEK, we specialize in high-performance lab equipment, including advanced presses essential for preparing consistent, high-quality samples for XRF and other analytical techniques.
Ensure your sample preparation meets the highest standards. Contact our experts today to discuss how our solutions can enhance your analytical workflow and deliver the reliable results you need.
Visual Guide
Related Products
- Lab XRF Boric Acid Powder Pellet Pressing Mold for Laboratory Use
- Automatic Laboratory Hydraulic Press for XRF and KBR Pellet Pressing
- XRF KBR Steel Ring Lab Powder Pellet Pressing Mold for FTIR
- XRF KBR Plastic Ring Lab Powder Pellet Pressing Mold for FTIR
- Laboratory Hydraulic Pellet Press for XRF KBR FTIR Lab Press
People Also Ask
- What are the differences between manual and automatic XRF pellet presses? Choose the Right Press for Your Lab's Needs
- What are specialized XRF pellet preparation presses designed for? Boost Lab Efficiency with High-Throughput Automation
- Why is the thickness of a pressed pellet important for XRF analysis? Achieve Infinite Thickness for Precise Results
- Why are pellets used in XRF analysis, and what is their limitation? Boost Accuracy and Speed in Your Lab
- What should be included in a checklist for making XRF pellets? Ensure Accurate and Repeatable XRF Analysis










