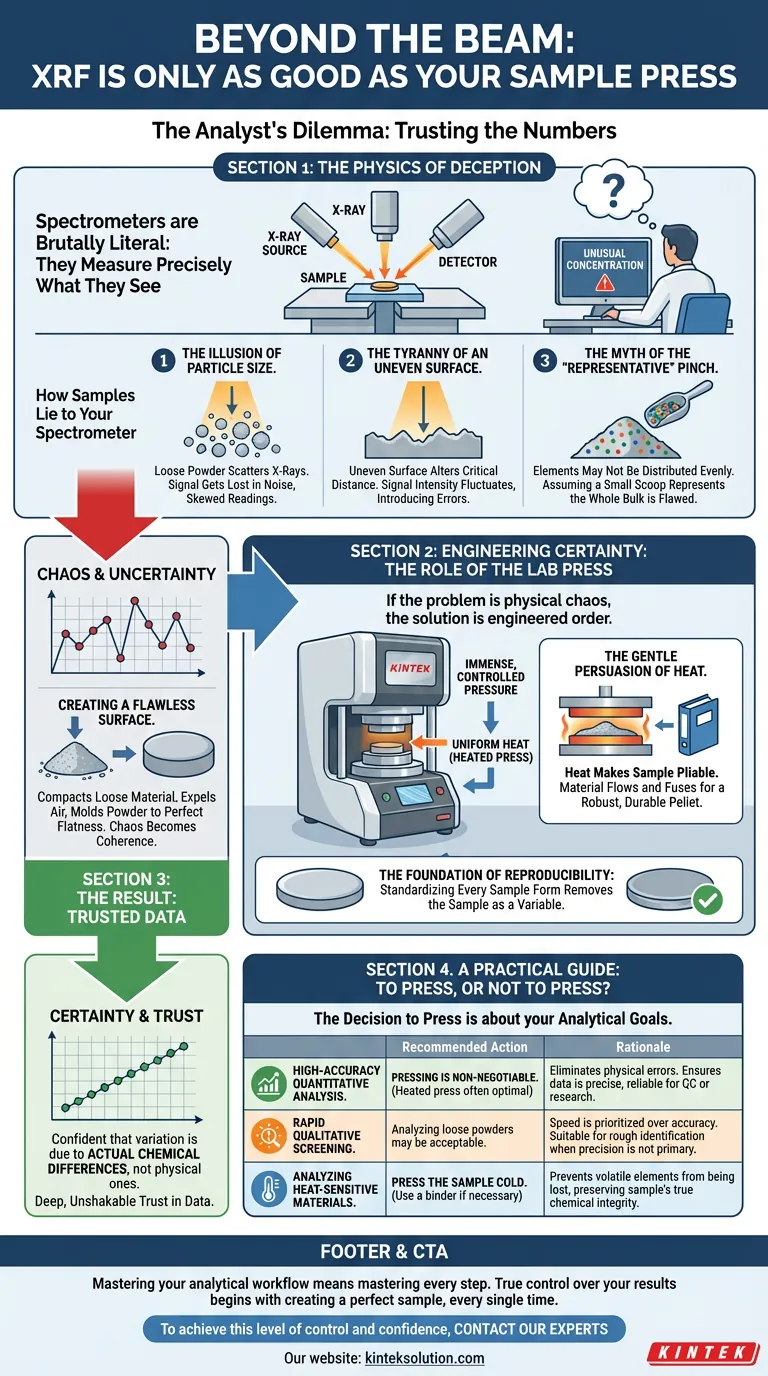
The Analyst's Dilemma: Trusting the Numbers
An analyst stares at a screen. The X-ray fluorescence (XRF) spectrometer reports an unusual elemental concentration in a geological sample—a result that could derail a major mining survey. The immediate question isn't about the element, but about belief: Is the data real, or is it an artifact?
This moment of doubt is familiar to anyone in analytical science. We place immense faith in our instruments, but spectrometers are brutally literal. They measure precisely what they see. If they see a loose powder with an uneven surface, they report the chaotic physics of that surface, not just the pure chemistry within.
The machine isn't lying. The sample is simply misleading it.
The Physics of Deception: How Samples Lie to Your Spectrometer
An unprepared sample presents a series of physical illusions that corrupt chemical data. XRF is a surface technique, making it exquisitely sensitive to the sample's form. To get to the truth, we must first understand the lies.
The Illusion of Particle Size
Imagine trying to determine the true color of a beach by looking at a handful of sand. The countless tiny surfaces scatter light in every direction, creating a washed-out appearance. A loose powder does the same to X-rays.
This particle size effect causes X-rays to scatter unpredictably between grains. The signal from lighter elements can get lost in the noise, while the signal from heavier elements can be artificially amplified. The result is a skewed, unreliable reading.
The Tyranny of an Uneven Surface
XRF instruments are calibrated for a perfect geometric relationship between the X-ray source, the sample, and the detector. An uneven surface, even on a microscopic level, alters that critical distance.
It's like a portrait photograph being slightly out of focus. Parts of the surface are too close, others too far away. The signal intensity fluctuates, introducing errors that have nothing to do with chemical composition. The machine assumes a perfect plane, and our sample violates that assumption.
The Myth of the "Representative" Pinch
We take a small scoop of material and assume it represents the whole batch. But elements may not be distributed evenly. While pressing doesn't change the microscopic makeup, it forces the material into a dense, homogenous disc. This ensures the surface seen by the X-ray beam is a true and fair representation of the bulk sample, turning an assumption into a physical fact.
Engineering Certainty: The Role of the Lab Press
If the problem is physical chaos, the solution is engineered order. A lab press doesn't just flatten a sample; it systematically removes the physical variables that create analytical doubt. It forges certainty.
Creating a Flawless Surface
The primary action of a lab press is applying immense, controlled pressure. This force compacts loose material, expels air, and molds the powder against a polished die. Chaos becomes coherence. The result is a dense pellet with a perfectly flat and smooth surface—the ideal canvas for X-ray analysis.
The Gentle Persuasion of Heat
Force alone is not always enough. This is where the elegance of a heated lab press comes in. Heat, applied through precisely controlled platens, makes the sample more pliable.
When mixed with a binding agent (like wax), heat allows the material to flow and fuse together, creating a robust, durable pellet that won't crumble or crack. It's not brute force; it's a calculated application of energy to achieve a more perfect and stable form. Instruments like KINTEK's heated lab presses are engineered for this exact purpose, providing the uniform temperature and pressure needed to build superior samples.
The Foundation of Reproducibility
By standardizing every sample into the same physical form—for example, a 40mm pellet—the press removes the sample itself as a variable. Now, when readings differ between two samples, we can be confident the variation is due to actual chemical differences, not physical ones.
This builds a deep, unshakable trust in the data. It transforms analysis from a series of isolated measurements into a reliable, comparable process.
A Practical Guide: To Press, or Not to Press?
The decision to press a sample is a decision about your analytical goals.
| Analytical Goal | Recommended Action | Rationale |
|---|---|---|
| High-Accuracy Quantitative Analysis | Pressing is non-negotiable. A heated press is often optimal. | Eliminates physical errors to ensure data is precise, reliable, and suitable for QC or research. |
| Rapid Qualitative Screening | Analyzing loose powders may be acceptable. | Speed is prioritized over accuracy. Suitable for rough identification when precision is not the primary goal. |
| Analyzing Heat-Sensitive Materials | Press the sample cold. Use a binder if necessary. | Prevents volatile elements from being lost, preserving the sample's true chemical integrity. |
This framework highlights the need for versatile and reliable equipment. Whether your work demands the robust pellets from a heated press or the careful compaction of a cold press, having the right tool is paramount. KINTEK's range of automatic and manual lab presses provides this critical capability for any laboratory.
Mastering your analytical workflow means mastering every step. The most advanced spectrometer in the world is only as good as the sample you present to it. True control over your results begins with creating a perfect sample, every single time.
To achieve this level of control and confidence in your XRF analysis, you need equipment designed for the task. Contact Our Experts
Visual Guide
Related Products
- Automatic Laboratory Hydraulic Press Lab Pellet Press Machine
- Carbide Lab Press Mold for Laboratory Sample Preparation
- Automatic Laboratory Hydraulic Press for XRF and KBR Pellet Pressing
- XRF KBR Steel Ring Lab Powder Pellet Pressing Mold for FTIR
- Laboratory Hydraulic Pellet Press for XRF KBR FTIR Lab Press
Related Articles
- The Tyranny of the Millimeter: Why Geometric Precision Dictates Fluoroelastomer Truth
- The Architecture of Density: Why Pressure is the Foundation of Precision
- The Alchemy of Pressure and Heat: Mastering TP-RTM with Precision Hydraulics
- The Geometry of Fusion: Why Precision Hot Pressing is the Silent Architect of Bio-Composites
- The Thin Margin of Success: Engineering the Interface in High-Temperature Composite Pressing




















