You’ve spent days, maybe weeks, perfecting your process. The component comes out of the hot press looking flawless—a smooth, solid, perfectly formed part. But then, during quality testing or its first real-world use, it happens: a sudden, catastrophic failure. The layers delaminate, the electronic joint gives out, or the bond simply splits apart under a fraction of its expected load. You're left holding two pieces, wondering what on earth went wrong.
If this scenario feels painfully familiar, you are not alone.
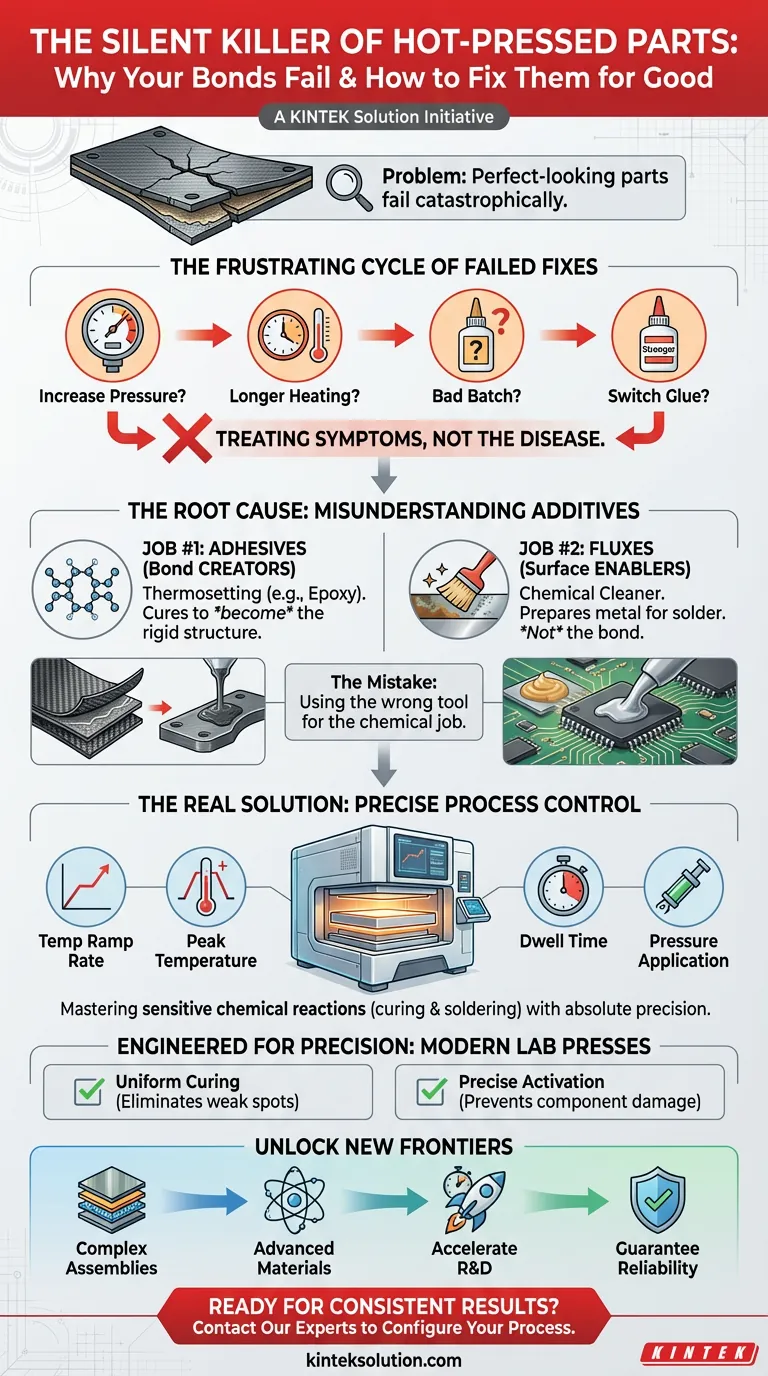
The Frustrating Cycle of Failed Fixes
When a hot-pressed bond fails, the troubleshooting scramble begins. The typical response is to address the most obvious variables:
- "Let's increase the pressure." The thinking is that more force will squeeze out any imperfections.
- "We need a longer heating cycle." Maybe the materials just didn't have enough time to "cook."
- "This batch of adhesive must be bad." It's easy to blame the raw materials and order a new supply, losing time and money.
- "We'll switch to a 'stronger' glue." A different product might solve the problem, right?
Sometimes, these tweaks produce a temporary improvement. More often, they lead to inconsistent results and more confusion. This isn't just a technical headache; it has serious business consequences. Every failed batch represents wasted high-value materials, blown R&D budgets, and critical project delays. For products that make it to market, a latent bond failure can lead to costly recalls and irreparable damage to your company's reputation.
The cycle continues because these "fixes" are just shots in the dark. They are attempts to treat the symptoms without ever diagnosing the real disease.
The Root Cause: Are You Creating a Bond or Just Cleaning the Surface?
The turning point comes when you realize that most bonding failures aren't caused by a lack of force or time. They're caused by a fundamental misunderstanding of the additives being used.
Many engineers use terms like "glue" or "flux" interchangeably, thinking of them as general-purpose "helpers" for sticking things together. In reality, they perform two completely different, and mutually exclusive, jobs. The secret to a perfect bond is knowing which job you need done.
Job #1: Adhesives as Bond Creators
In technical applications, "glue" is typically a thermosetting adhesive (like an epoxy or resin). This material isn't just a sticky filler. Under heat and pressure, it undergoes an irreversible chemical reaction called curing. It transforms from a liquid or putty into a rigid, structural solid.
- Its Role: To become the bond itself.
- Example: When creating a carbon fiber composite part for an aerospace application, layers of carbon fabric are infused with epoxy resin. The hot press provides the precise heat to trigger the curing reaction and the pressure to ensure a dense, void-free final structure. The cured epoxy is the matrix holding the fibers together.
Job #2: Fluxes as Surface Enablers
A flux, on the other hand, is a chemical cleaning agent. It is not an adhesive. Its only job is to prepare metal surfaces for joining with a filler metal, like solder.
Most metals instantly form a thin, invisible layer of oxide when exposed to air. This oxide layer acts like a barrier, preventing molten solder from making a true, atom-to-atom connection with the base metal. When heated, flux becomes chemically active and aggressively dissolves these oxides.
- Its Role: To prepare the surface so a different material (solder) can create the bond.
- Example: In electronics manufacturing, solder paste (a mix of solder spheres and flux) is used to attach a chip to a circuit board. During hot pressing, the flux activates, cleans the metal pads, and is then displaced by the melting solder, which forms a clean, strong metallurgical connection.
Here’s why the common fixes fail: Increasing pressure won't help if your real problem is an oxidized metal surface that the flux never cleaned. Likewise, changing adhesives is pointless if your goal is to solder two metals together. You're using the wrong tool for the job.
The Real Solution: Moving from Brute Force to Process Control
Understanding the difference between creating and enabling a bond is the first step. The second, and most critical, is executing that specific chemical process with absolute precision.
Both adhesive curing and flux activation are highly sensitive chemical reactions. They don't respond to brute force; they demand precise control over the entire process. To get a reliable, repeatable bond every single time, you need a tool that can flawlessly manage:
- Temperature Ramp Rate: How quickly or slowly you heat the part.
- Peak Temperature: Reaching the exact temperature to activate the reaction without damaging the components.
- Dwell Time: Holding that temperature for the precise duration needed for the reaction to complete.
- Pressure Application: Applying the right amount of force at the right time to ensure intimate contact and part integrity.
Designed for Precision: The Role of the Modern Lab Press
This is precisely why KINTEK laboratory presses are engineered as sophisticated process-control instruments, not just simple heating and squeezing machines. They are designed from the ground up to master the chemistries of modern bonding.
- For Adhesive Curing: Our automatic heated presses provide exceptionally uniform platen temperatures and programmable multi-stage cycles. This ensures that every square millimeter of your adhesive cures completely and evenly, eliminating the weak spots that lead to delamination.
- For Flux-Based Soldering: The ability to precisely control the temperature profile means you can activate the flux at the perfect moment to clean the surfaces, followed by the exact temperature spike needed to reflow the solder—all without overheating and damaging sensitive electronic components.
By replacing guesswork with programmable precision, you eliminate the primary variable that causes bonding failures: inconsistent process control.
Beyond Reliable Bonds: What New Frontiers Can You Explore?
When you can finally trust your bonding process, a fundamental shift occurs. You stop wasting time and resources troubleshooting basic failures and start unlocking new potential.
Instead of re-running failed experiments, your R&D team can confidently:
- Develop more complex multi-material assemblies, combining ceramics, metals, and polymers.
- Test and qualify advanced, high-performance materials that require narrower processing windows.
- Accelerate product development cycles, moving from prototype to production faster than ever before.
- Guarantee the reliability of mission-critical components in fields from aerospace to medical devices.
You move from a state of uncertainty to one of control, turning your hot pressing capability from a source of frustration into a true competitive advantage.
Your bonding challenges are unique, and the solution should be too. If you're ready to move past recurring failures and achieve consistent, reliable results in your lab, our team is here to help you configure the right process. To discuss your specific project needs and how precise process control can transform your outcomes, Contact Our Experts.
Visual Guide
Related Products
- Lab Heat Press Special Mold
- Warm Isostatic Press for Solid State Battery Research Warm Isostatic Press
- Lab Anti-Cracking Press Mold
- Lab Infrared Press Mold for Laboratory Applications
- Automatic Heated Hydraulic Press Machine with Heated Plates for Laboratory
Related Articles
- The Architecture of Pressure: Why Precision is the Only Truth in Material R&D
- The Physics of Transparency: Mastering the Matrix in FTIR Starch Analysis
- The Last Millimeter: Why Physical Constraints Define Material Integrity
- The Invisible Architect: Engineering Structural Harmony with the Heated Press
- The Silent Symmetry: Why Homogeneity is the Soul of the Pressed Pellet




















